This specimen caused KT to have to do some research on both Mindat.org and elsewhere! First, KT's personal Royal Efforts. His Majesty spotted this piece on eBay at least 3 weeks ago, and wanted it badly as it displayed a texture which He was most interested in while in university, Gosh over 50 years ago! HA HA. Well, someone outbid me and I figured it is gone as it sold. Then a couple of days later I get an email from the seller saying it had been relisted if His Majesty was still interested. And the relisted price had dropped a bit, so KT put in a bid for what my high bid was before, held my breath for 4 days, and won it!
About the specimen itself, it is a nearly half inch thick slab of coarse grained calcite, zincite, tephroite, and franklinite, from Franklin Mine, Sussex Co., NJ. KT is excited to see it fluoresce because He was interested in the texture of the willemite in this specimen and that it is mostly confined to the mineral tephroite.
KT refers you to the following article in American Mineralogist, V 70, pgs. 568-575, 1983 by Carl A. Francis, New Data on the Forsterite-Tephroite Series. Tephroite is the Manganese end member within the Olivine Group. Francis worked on Tephroite from the well known zinc deposits of Sterling Hill and Franklin, NJ. He found that Zinc-bearing olivines occur in the metamorphosed Zn-Mg-Fe deposits there. Individual crystals of Tephroite contain oriented lamellae of willemite and he considered that strong evidence of exsolution origin for the willemite of that association. He also found that Tephroite originally contained ~20 vol. % Zn2SiO4 components.
Exsolution as defined is a process whereby an initially homogenous solid solution (a crystal, KT) separates into 2 or more distinct crystalline phases (minerals, KT) without change to the bulk composition (chemistry, KT). (Source: AGI, Mindat.org). No addition or deletion of material is necessary. It is a process whereby a mineral that formed at high temperatures will separate mechanically into two distinct minerals, even when it is a solid. Why does this happen? Because at high temperatures, the lattice structure of the initial mineral is more open and can accommodate variously sized atoms and cations, but as the mineral cools, the structure collapses, and those cations and atoms that are now too large to stay in that structure, so they separate and form a different mineral. When you look at the overall chemistry, there is no change!
We see this most commonly in the Feldspar Group, particularly the plagioclases (perthites and antiperthites) and in microcline (as tartan patterns in said minerals), but it is also known to occur in high temperature ilmenite where the final two minerals are magnetite and hematite. Also, well known from the change of high temperature Leucite to a mixture of nepheline and k-feldspar. This is for me the first time I have seen it in tephroite as that mineral is pretty scarce worldwide. This specimen is from the type locality (TL) for tephroite.
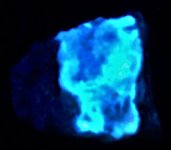
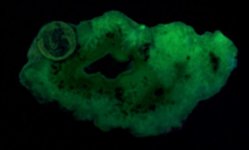
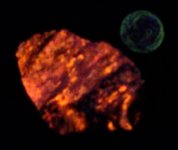
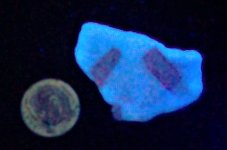
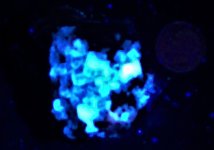
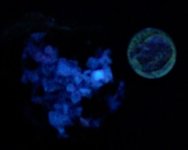
Anyway, here are a series of photographs, the first two with a US dime for size comparison. The gray matrix is calcite, the black opaque is franklinite (TL), the orangish red is zincite, and the brown blebs are tephroite in the first image. Second picture shows the specimens response to SW 245nm light....calcite varies from orange to yellowish, and willemite is green.
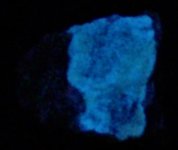
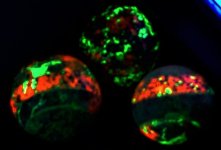
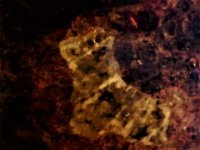
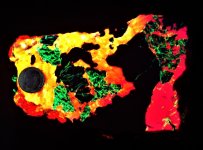
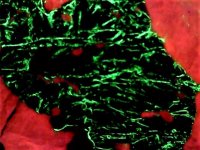
The second set of images was taken at 10X with my USB Chinese led toy microscope and the first picture in natural light shows a homogenous appearing coarse grain of tephroite set in calcite. But the second picture of this set shows linear streaks and sets of fluorescent green willemite within the same grain. Again the lamp is SW 245nm. The willemite is either as thin streaks or veinlets and as apparent fracture fillings within the tephroite grain. Very little of the willemite is within or cutting through the other minerals present...as revealed in the 2nd image of the set.
Hope you find this info and specimen as interesting as I do! HA HA